You are here
News
2024
May 2024 StemCyte's CEO explains why the company is ready for an IPO. In addition to operating a multi-national hybrid cord blood bank, StemCyte is also manufacturing immunotherapies from cord blood and is one of the industry's leaders in running clinical trials with cord blood.
May 2024 For generations, Indians have stored a baby's dried umbilical cord in an amulet. The new practice of cryopreserving living perinatal stem cells builds on these old customs. Marketing of family cord blood banking is very personalized in India, where sales representatives visit families in their homes.
Apr 2024 Exclusive: How a small group of patient advocates and biotech companies tricked the Utah legislature into passing a law which sets a precedent. Utah is now the first state to legalize therapy with stem cells from placenta tissue and cord blood.
Apr 2024 Parents are always told that there are no biomarkers for diagnosing Autism. But they are not told that over a thousand biomarkers have been studied. To satisfy the American Psychiatric Association, a proposed biomarker would have to consistently be over 80% accurate. One of the biggest challenges in identifying biomarkers is validating them against the existing behavioral scores for autism, which do not consistently meet the 80% standard.
Mar 2024 The purpose of the National Cord Blood Network is to promote the use of cord blood grafts and products to transplant doctors, and to thereby improve transplant access for patients from underserved populations. The audience of the National Cord Blood Network is the medical community at US transplant centers. The authority of the National Cord Blood Network comes from their participating hematologists and oncologists that provide guidance and training.
Mar 2024 Gabriel and Alex received their own Cord Blood as therapy for Autism. Neither boy has been "cured" of Autism, but both have had remarkable gains.
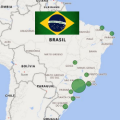
Feb 2024 Brazil has a big public health investment in a national network of 14 public cord blood banks. But the medical community in Brazil is not actively utilizing this resource or developing new therapies that employ donated cord blood.
Feb 2024 New research finds overall survival is better after haplo transplants when patients also receive a small infusion of partially matched cord blood. This protocol might lead to enhanced utilization of the smaller cord blood units in public banks.
Jan 2024 Mesenchymal Stromal Cells (MSC) have been bioprinted into a human knee meniscus in zero gravity for the first time. If human organs can be bioprinted in space, they will satisfy an unmet need on Earth where patients are waiting for transplants. They will eventually be part of the medical toolkit for long-haul astronauts seeking to colonize planets.
Jan 2024 FDA rules allow for a blood donor to receive a payment in the form of a gift card, with no limit on the type of card or the amount. This makes it possible for expectant moms to receive large gifts in exchange for cord blood donations. So long as the payment is in the form of a gift card, these donations are still labelled "volunteer donor" and can be used to treat patients.