You are here
Milestones
| 1966 |
| Doctors Milton and Norman Ende were brothers that published the first case of cord blood as cancer therapy. The Ende brothers are often overlooked in official histories of cord blood transplantation because they did not use immune suppression before treating patients with cord blood, yet their publications demonstrated transient efficacy of cord blood alone. Until his death at age 96 in 2021, Dr. Norman Ende was a passionate advocate for delayed cord clamping. |
| Oct. 1988 |
| The Founders of Cord Blood Banking who made the world's first cord blood transplant possible were: Matthew Farrow, the 5 year old patient with Fanconi Anemia, Dr. Joanne Kurtzberg, Matt's doctor in the US, Dr. Arlynn Auerbach, who checked the genetic match between Matt and his baby sister, Dr. Gordon Douglas, the obstetrician who delivered the baby, Dr. Hal Broxmeyer, the scientist who preserved the cord blood and transported it to the hospital, and Dr. Eliane Gluckman, the doctor who performed the transplant at the Hôpital Saint-Louis in Paris, France. |
| May 1989 |
| A team led by Dr. Edward A. Boyse, including Dr. Hal Broxmeyer, published in the Proceedings of the National Academy of Sciences that it was feasible to collect umbilical cord blood from birth, ship it to a lab, and cryopreserve it for later therapy. |
| Sept. 1989 |
| Natalie Curry received one of the early cord blood transplants with Dr. Gluckman to treat Fanconi Anemia. Natalie's sister Emily was her cord blood donor, and later when Emily turned age 18 she also donated a kidney. Natalie went on to become a vocal advocate for cord blood education. |
| Jan. 1992 |  | Dr. Pablo Rubinstein spearheaded the founding of the world's first and largest public cord blood bank at the NY Blood Center. Dr. Rubinstein is also credited with pioneering methods of cord blood collection and processing that became industry standards. |
| 1993 |  | Mitch Santana became the first person to receive a cord blood transplant from an unrelated donor when he was less than 2 years old. He was cured of acute leukemia thanks to an anonymous baby whose cord blood had been donated to the NY Blood Center in its first year of operation. Dr. Kurtzberg performed the transplant at Duke Medical Center. |
| Feb. 1995 |
| Dr. Mary Laughlin performed the world's first cord blood transplant for an adult leukemia patient. She was then at Duke Medical Center, but went on to found transplant programs at the Cleveland Cord Blood Center and the University of Virginia. |
| August 1996 |
| Dr. Mitchell Cairo and Dr. John Wagner led the first demonstration that long term patient survival is comparable with cord blood transplants and bone marrow transplants. Numerous researchers have reinforced this conclusion over the years, both for children and adult patients. ref1, ref2 |
| Nov. 1997 | Stephen Sprague was the first adult to receive an "expanded" cord blood transplant where the cells were first grown in the lab before infusion. He was treated for CML leukemia in blast crisis by Dr. Andrew Pecora at the Hackensack Medical Center. | |
| June 1998 |
| Dr. Frances Verter founded the Parent's Guide to Cord Blood website in memory of her daughter Shai. This photo was taken at a playground in Brooklyn NY in 1994. |
| Dec. 1998 |
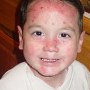
| Keone Penn was the first person cured of sickle cell disease by a cord blood transplant. As a child, he had endured a stroke and frequent episodes of pain before the transplant at Egleston Children's Hospital in Atlanta. Dr. Andrew Yaeger found a matching cord blood donor for Keone in the NY Blood Center public bank. |
| Apr. 2001 |
| Shortly after he was born, Jesse was diagnosed with eye cancer that had invaded both eyes and his spinal fluid. His treatment at the Hospital for Sick Children in Toronto, Canada, culminated with the infusion of his own cord blood that had been stored with Insception Lifebank. Jesse was one of the first children who were saved because their parents had banked their cord blood privately. |
| 2002 |
| Patrizia Durante was diagnosed with leukemia as she was starting the third trimester of her first pregnancy. Her daughter Victoria Angel was delivered early so that Patrizia could start chemotherapy. Later, Patrizia was cured of her leukemia after a transplant of her child's cord blood. Their story inspired the formation of a cord blood donation program called the Victorial Angel Registry of Hope, which operated in Toronto as a subsidiary of Cells for Life family bank. |
| September 2002 | Dr. John Wagner published a key study in the journal Blood that analyzed the influence of stem cell dose and degree of donor-patient match on the outcome of cord blood transplants. There have been many subsequent papers analyzing this topic. ref | |
| Apr. 2004 |
| Andrej was 6 months old when he became the first child to receive a transplant of his own cord blood to cure a malignant brain tumor called medulloblastoma. His cord blood had been stored with Eurocord-Slovakia, a partner of the family bank group Cord Blood Center. The treatment took place at the University Children's Hospital in Bratislava, Slovakia. |
| Aug. 2004 |
| Dr. Ammar Hayani and colleagues performed the first autologous cord blood transplant for acute leukemia. Typically, the mutation(s) that cause childhood leukemia are present since birth, and the patient cannot be cured by their own cord blood. In this published case, genetic testing revealed that the leukemia was an acquired mutation. The 3 year old girl's cord blood had been stored in the bank CorCell (now owned by Vitalant New Jersey), and the transplant took place at Advocate Hope Children's Hospital. |
| Feb. 2005 |
| Abby Pell was the first child to receive her own cord blood stem cells, banked at Cord Blood Registry, as experimental therapy for brain injury. Abby suffered from oxygen deprivation at birth. Her mother had been turned away by doctors at multiple hospitals before Dr. Kurtzberg agreed to try the procedure at Duke Medical Center. |
| May 2005 |
| Dr. Joanne Kurtzberg and colleagues published the first paper demonstrating that cord blood transplants can stop neurologic damage from metabolic storage disorders, by helping patients to create the metabolic enzymes they lack. Their initial work was with patients who had Krabbe's disease and appeared in the New England Journal of Medicine. Her group went on to treat dozens of other rare metabolic disorders. |
| Oct. 2005 |
| Ryan Schneider was the first child diagnosed with cerebral palsy to be treated with his own cord blood. His cord blood had been stored with Cord Blood Registry and he was treated by Dr. Joanne Kurtzberg at Duke Medical Center. Ryan's mother Mary Schneider went on to become a cord blood advocate, lobbying tirelessly in support of legislation that would increase cord blood education, research, and access to therapies. |
| Oct. 2007 |
| Jake Liao was the first child to receive a cord blood transplant for a rare and fatal skin disorder called epidermolysis bullosa, or EB. The discovery that stem cell transplants could teach the body to produce missing skin proteins was made at the University of Minnesota, by a team led by Dr. John Wagner and Dr. Jakub Tolar. |
| Mar. 2008 |
| Dr. J.J. Nietfeld led a team of researchers that published an analysis of U.S. stem cell transplant statistics. Over the course of childhood up to age 20, only 3 in 5,000 people will have a stem cell transplant. But over the course of a 70 year lifetime, the number of people that have a stem cell transplant (not just need one, but have one) is 0.46%. Thus the lifetime probability of a stem cell transplant is 1 in 217. |
| 2008 |
| Starting in 2008, Brazil’s National Cancer Institute (INCA) invested about R$ 3.5 million per bank to establish a national network of 14 public cord blood banks. Transplant doctors told mothers that they should donate their cord blood, not store it privately. Yet, by the 2020’s cord blood had fallen out of political favor in Brazil, and by the end of 2023 none of the public banks are accepting donations. Now the only cord blood banks serving parents in Brazil are the family banks. |
| Sep. 2010 |
| Dr. Jessica Sun and colleagues published a paper describing the therapies given to the first 184 children with acquired neurological disorders who received their own cord blood at Duke Medical Center. These patients had diagnoses including cerebral palsy, hydrocephalus, oxygen deprivation at birth, hypoxic-ischemic encephalopathy, stroke, etc. |
| Mar. 2011 |
| Dr. Charles S. Cox Jr. and colleagues published in the journal Neurosurgery that giving children their own stem cells showed promise as a treatment for traumatic brain injury, the leading cause of death in children. Dr. Cox is a neurosurgeon who directs the pediatric trauma program at the University of Texas Medical School in Houston. |
| Nov. 2011 | Ever since she performed the first cord blood transplant in 1988, Dr. Eliane Gluckman has led studies that compared the outcome of cord blood transplants with related versus unrelated donors, always finding that related are better. Her publication in 2011 found transplant survival 1.5 times better with sibling donors. | |
| August 2012 |
| Dr. Michael Chez, director of pediatric neurology at the Sutter Neuroscience Institute in Sacramento, launched the first clinical trial to treat children who have autism with their own cord blood. According to the CDC, the number of U.S. school children on the autism spectrum doubled over the decade from 2006 to 2016, from 1 in 110 to 1 in 54. ref1, ref2 |
| Dec. 2012 |
| Dr. MinYoung Kim and colleagues at CHA Bundang Medical Center in South Korea published the first demonstration that cord blood therapy is effective for cerebral palsy. The efficacy was measured by improvements in motor skills scores in a placebo-controlled, double-blind clinical trial. |
| Dec. 2012 |
| Parent's Guide to Cord Blood reports that, worldwide by the end of 2012, family banks had released over 900 cord blood units for therapy. Overall, roughly half went to sibling transplants and half were used by the child from which the blood was banked. |
| April 2013 | Dr. John Wagner performed the world's first cord blood transplant of a child who had both leukemia and HIV. The goal was to cure both by transplanting cord blood from a donor who carries a rare genetic mutation that confers resistance to HIV. Dr. Wagner heads the University of Minnesota's transplant program. ref1, ref2 | |
| July 2013 |
| Dr. Karen Ballen, together with Dr. Gluckman and Dr. Broxmeyer, published a review of the first 25 years of cord blood transplantation in the journal Blood. To date, more than 30,000 cord blood transplants have been performed around the world. |
| Aug. 2017 |
| In Feb. 2022 it was announced that an anonymous female patient with HIV was cured by a cord blood transplant that had taken place in August 2017, at Weill Cornell Medical Center in New York City. This treatment is not destined to become routine: A stem cell transplant can only cure HIV if the donor carries a mutation called CCR5-Δ32. This mutation is rare, and stem cell transplants are sufficiently risky that they are only offered to AIDS patients that develop leukemia. The first HIV patient cured by a stem cell transplant, using bone marrow, was the "Berlin patient" Timothy Ray Brown (pictured) in 2007. |
| Oct. 2017 |
| Duke University has received FDA approval for an Expanded Access Protocol that allows cord blood infusions (autologous or sibling) to treat acquired brain disorders. In the following years, the waiting list for this EAP program has grown to thousands of children. |
| Nov. 2018 |
| Parent's Guide to Cord Blood survey finds that the world inventory of cord blood units in storage is now about 750 thousand in public banks and nearly 7 million in family banks. The top 10 cord blood banks hold over 60% of the world's privately stored cord blood. |
| March 2019 |
| Dr. Juliet Barker of Memorial Sloan-Kettering Cancer Center is the leading expert on the use of double cord transplants to treat adults. She has argues that cord blood transplants remain a vital resource for patients that are not represented on bone marrow registries and/or possess rare genetic types. ref1, ref2 |
| Sep. 2019 |
| Dr. Ilya Yemets, together with colleagues at the Center for Pediatric Cardiology in Kyiv Ukraine, has performed over 100 cardiac surgeries in which children with severe congenital heart defects received their own cord blood to support their cardiopulmonary bypass. |
| June 2020 |
| The NY Blood Center suddenly shut down their famous cord blood donation program, ceasing all collections and letting go all research staff. A small crew continues to work in the laboratory to maintain the inventory and release units called for transplant. Both current and former employees are forbidden to speak of this shut down. |
| Aug. 2021 |
| It is well known that childhood leukemia is caused by mutations that were present at birth. But it is less well known that the mutations are not predictive: some mutations occur in a thousand more children for every one that is diagnosed with leukemia. Drs. Logan Spector (pictured) and Adam de Smith at the University of Minnesota are comparing children's mutations at the time of leukemia diagnosis versus mutations in their cord blood to better understand what triggers the onset of leukemia. The ultimate goal is to be able to predict and prevent childhood leukemia. |
| Dec. 2022 |
| Duke University Medical Center has voluntarily excluded Autism from its Expanded Access protocol. This decision was a reaction to a campaign of negative publicity. |
| Mar. 2023 |
| Dr. Masoumeh Nouri, of Royan Institute for Stem Cell Biology and Technology in Iran, led the first study of how cerebral palsy responds to cell therapy as a function time. The study compared 36 patients that received cord blood stem cells, versus 36 patients that received cord tissue MSC (Mesenchymal Stromal Cells), versus a control group of 72 patients. The comparison found that while initial response was stronger with cord blood stem cells, by the end of a year there was no significant difference between the two cell therapy groups. |
| 2023 |
| In 2023, beauty pageant contestant Sosa Evbuomwan shared that she was the first child cured of sickle cell disease by a transplant of the expanded cord blood product from Gamida Cell (see next milestone). |
| Apr. 2023 |
| Gamida Cell achieved the first FDA approval for transplants with an expanded cord blood product. The cord blood is expanded with the molecule nicotinamide, and the product name is Omisirge (previously known as Omidubicel and NiCord). This expanded product engrafts in 10-11 days, faster than bone marrow, but takes four weeks to manufacture. |
| May 2023 |
| A team at the Royal Manchester Children’s Hospital, led by Dr. Robert Wynn, performed a retrospective study of pediatric stem cell transplants in the UK & Ireland. They found that for those leukemia patients going into transplant with minimal residual disease (MRD), cord blood transplants yielded dramatically better survival than other graft sources. Their research into the mechanism of action suggests that the T-cells in cord blood carry a potent graft-versus-leukemia effect that is missing in transplants from adult donors, especially haploidentical (Haplo) bone marrow transplants where T-cells are suppressed. |
| July 2023 |
| Catching up with Matt Farrow at age 40: In this interview Matt talks about being one of the oldest survivors living with Fanconi Anemia, and the pros and cons of being a legend in the cord blood arena. His donor baby sister gave birth to her second child in 2023 and stored the cord blood with MiracleCord. |
| Sep. 2023 |
| Dr. Hal Broxmeyer, known to many of us as the “Father of Cord Blood Transplantation”, was the scientist that invented our standard protocols for cryopreserving cord blood. His laboratory has demonstrated that cord blood can be frozen for 27 years without any diminishment in the yield of viable stem cells. |
| Oct. 2024 |
| Cord Blood use in China rivals or exceeds the rest of the world combined. Western researchers count 40,000 cord blood transplants to date. This number covers the world except for China. Meanwhile, the Chinese research community counts 40,000 therapeutic applications of cord blood to date in China alone. |
| Dec. 2024 |
| Dr. Colleen Delaney has been a leader in the development of Natural Killer (NK) cell therapies sourced from cord blood. She helped found the company Deverra Therapeutics to develop the NK product Dilanubicel. In a recent study of 15 patients with high-risk leukemias that were followed for one year, the disease-free survival was 100% and the rates of acute or chronic graft versus host disease were zero. |
| Dec. 2024 |
| Dr. Vanderson Rocha is a hematologist who worked with Dr. Eliane Gluckman in Paris, publishing over 200 papers. He has had prestigious appointments at many research centers. In 2016 he returned to his native Brazil as Full Professor of Hematology at the University of São Paulo, and President of the Pró-Sangue Foundation, the largest public blood bank in Latin America. During 2025, Dr. Rocha has been fighting his own cancer, and he has talked candidly about how being a patient has changed his perspective. |
| Mar. 2025 |
| Dr. Qifa Liu is the Director of Haematology at Nanfang Hospital in Guangzhou, China. In Chinese hospitals, cord blood stem cells are often given as a supplement to other forms of stem cell transplants. Dr. Qifa Liu's group performed a phase 3 multi-center trial that tested Haplo-Cord transplants against Haplo transplants for haematological malignancies. The results, published in the journal Lancet, found that Haplo-Cord transplants were significantly better (p=0.001) in terms of disease-free survival. |
| Mar. 2025 |
| The journal Pediatrics published a study which uses a type of statistics called IPDMA to merge data from 498 participants in 11 previous studies of cord blood therapy for cerebral palsy. This research was led by the international Cerebral Palsy Alliance and the first author was Dr. Megan Finch-Edmondson (pictured). The results proved that cord blood therapy has a large treatment benefit for the motor skills of cerebral palsy patients (p=0.012).Additional trends found: younger children benefited more, higher functioning patients benefited more, and bigger cell doses were better. Cerebral palsy is the most common motor disability of childhood. |
| Apr. 2025 |
| Zara was the first child in Australia to receive her own cord blood as therapy for cerebral palsy under a new "special access scheme". Zara's cord blood had been stored at the bank Cell Care, and she was treated under the supervision of Dr. Michael Fahey, pediatric neurologist at Monash Children’s Hospital. |
| Aug. 2025 |
| ExCellThera has obtained EU approval for their expanded cord blood product Zemcelpro. The cord blood is expanded with the proprietary molecule UM171. This expanded product engrafts in 20 days, and it can be manufactured in only one week. |
| Sep. 2025 |
| The Rainbow of Heroes Walk celebrates its 25th anniversary. This event brings together survivors who received stem cell transplants at the Duke division of Pediatric Transplant and Cellular Therapy. It is moving to meet multiple young adults who received a cord blood transplant over two decades ago. |
| Oct. 2025 |
| The 2025 Nobel Prize in medicine was awarded for discovering the role that regulatory T-cells (T-regs) play in immune tolerance. Numerous companies are developing therapies with T-regs to combat auto-immune diseases. Cord blood is the richest source of T-regs, a type of immune cell whose concentration in blood declines with age. Moreover, the T-regs in cord blood have minimal exposure to antigens, and cord blood T-regs are better at maintaining their properties when expanded in culture. The company Cellenkos, founded by Dr. Simrit Parmar (pictured), published in NEJM that T-regs from cord blood slowed the progression of ALS (amyotrophic lateral sclerosis). |
| Jan. 2026 |
| FamiCord, the largest multi-national family cord blood bank in Europe, provides a program for their client families where a child can receive his/her own cord blood for brain conditions like cerebral palsy and autism. The treatments are conducted under a Hospital Exemption (HE, the EU version of Expanded Access) at the University Children’s Hospital in Lublin, Poland. In 2019 this program began providing cerebral palsy therapy, then Poland revised their regulations for HE during 2018-2023, and now the program is active again and includes autism. |