You are here
World's 1st Trial of Amnion Stem Cells for Premature Babies

The medical potential that lies in cord blood is clear and the cord blood banking industry is booming, but have you ever considered the remaining perinatal tissues that are routinely discarded as medical waste?
Walking through the aisles of some pharmacies or healthcare stores, you will find capsules of sheep placenta being sold as health supplements, "placental hair masks" and other such things. There are now international services that offer dehydration and encapsulation of your baby's placenta, even placenta cookbooks! A more realistic service is offered by a few cord blood banks that preserve the stem cells from placental blood and placental tissue.
Additional perinatal tissues also have value. The fetal membranes - amnion and chorion - also contain stem cells and stem cell-like cells that can be isolated and banked for clinical use. The amnion and chorion ("amnion" hereafter) form a membrane that surrounds the baby during pregnancy and plays an important role in protecting the baby from physical and immunological damage. (Motifolio credit for diagram below)
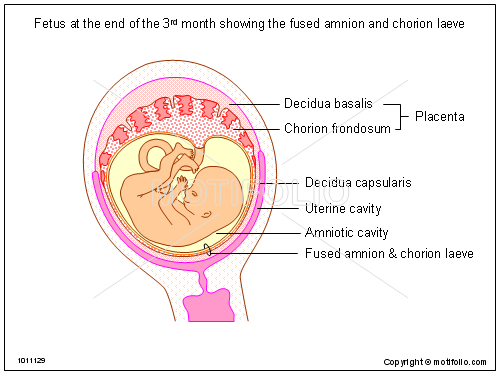 |
The amniotic membrane has unique wound healing properties and has been used as a wound dressing for decades. This is because the amnion has potent anti-inflammatory, pro-regenerative and scar-reducing properties. In fact, a number of randomised clinical trials favour the use of amnions in burns for their promotion of wound healing, patient comfort, and reduced need for dressing changes. The amnion dressings promote cell growth, modulate inflammation and recruit endogenous (local) stem cells to facilitate wound repair.
Perinatal stem cells and maternal tolerance
Research has provided insights into the complexity of the interaction between the mother's and baby's separate immune systems; this interaction takes place at the maternal-fetal interface and is necessary for a healthy pregnancy. One of the key changes in the immune system of an expectant mum is the switch from immune surveillance to immune tolerance. In healthy full-term pregnancies, this switch prevents the rejection of the fetus, which is essentially foreign to the mother's immune system.
The switch in a mother's immune system during pregnancy is largely mediated by regulatory T cells in the maternal bloodstream. We now know that some of the stem cells and stem-like cells of the placenta and fetal membranes have a vital role to play in this immunological switch. They are responsible for triggering cellular events such as activating the maternal regulatory T cells that maintain tolerance and promoting their proliferation, while concurrently inducing the death of activated T cells that could compromise the pregnancy.
Bankable cell types from perinatal tissues
Mesenchymal Stem Cells (MSCs) have been successfully isolated from first-, second-, and third-trimester placental compartments, including the amnion, chorion, decidua parietalis and decidua basalis. The MSCs represent less than 1% of the cells present in the placenta, while the average yield of MSCs from chorions and amnions is between 1 to 10 million per gram of tissue. Regardless of their source, MSCs have potent immunosuppressive properties and MSCs obtained from the fetal membranes are no exception. Chorionic and amniotic MSCs suppress T cell proliferation to the same extent as bone marrow derived MSCs.
Amnion Epithelial Cells (AECs) are a stem-like cell that can be isolated in large numbers (150 to 200 million per gram) from full-term placentae. Here are their technical properties: AECs have multipotent differentiation potential and expression of pluripotent stem cell specific transcription factors including Oct4 and Nanog. Unlike MSCs, which are all plastic adherent, primary AECs have three subpopulations of cells that are adherent, loosely adherent, and free-floating. Of these subpopulations, the adherent AECs have the lowest expression of Oct4 and Nanog. Notably, most in vivo studies to date have used the entire AEC fraction rather than a specific subpopulation of AECs.
AECs share many immunosuppressive properties with MSCs. They both release prostaglandins that mediate inflammatory response and regulate tissue repair. AECs also release HLA-G, a histocompatibility leukocyte antigen that can aid with tissue repair. The HLA-G molecule plays a major role in immune tolerance during pregnancy, particularly at the maternal-fetal interface, where HLA-G inhibits the migration and proliferation of maternal immune cells.
The future of perinatal stem cells
In summary, the placenta and other perinatal tissues are a rich source of stem cells and stem-like cells, which can potentially be banked in a similar manner to cord blood. Rather than banking only the cord blood and discarding the rest of the perinatal tissues, it is time we started routinely banking all of the perinatal tissues and the vast stores of stem cells and stem-like cells that they contain.
Researchers are working towards the translation of perinatal stem cells from the laboratory bench to the patient bedside. Both MSCs and AECs from perinatal tissues have been shown to exert potent immunomodulatory and pro-regenerative effects in animal models of bronchopulmonary dysplasia (BPD), a major health risk for severely premature infants. At the Ritchie Centre of Monash University in Australia, we are running the world's first clinical trial to assess the safety of amnion epithelial cells in premature babies with established BPD.


