You are here
Novel Treatment for Pancreonecrosis Using Cord Blood and Cord Tissue
 Acute pancreatitis is an acute inflammation of the pancreatic gland, a very severe condition that often results in the death (necrosis) of that part of the pancreas or even the whole organ. This in turn causes between 40% and 70% of patients with pancreonecrosis to die, and even in the world’s best surgical centres the death rate is not lower than 20%.
Acute pancreatitis is an acute inflammation of the pancreatic gland, a very severe condition that often results in the death (necrosis) of that part of the pancreas or even the whole organ. This in turn causes between 40% and 70% of patients with pancreonecrosis to die, and even in the world’s best surgical centres the death rate is not lower than 20%.
Acute pancreatitis ranks as the third most prevalent disorder of the abdominal cavity that requires urgent surgical intervention. The main causes of acute pancreatitis are alcohol abuse (seen in patients about 40 years old) and gallstones (in patients over 60 years old).
The pancreatic gland is an important organ as it produces a number of essential digestive enzymes and is involved in the regulation of blood sugar level, producing the hormones insulin and glucagon. In healthy patients, the digestive enzymes produced by the pancreatic gland are only activated in the digestive tract, whereas during pancreatitis the pancreatic gland digests itself, escalating the severity of the condition.
The treatment of acute pancreatitis requires the patient to fast and take drugs which inhibit the enzymes that digest protein, as well as drugs to relax muscle spasms, antibiotic drugs, and painkillers. Very often patients require surgery to remove the necrotic (dead) tissues of the gland. Patients also require the insertion of tubes to drain fluid in the abdominal cavity. Depending on the severity of the pancreonecrosis the patient may require multiple surgeries.
Due to the severity of this disease and the insufficient efficacy of established therapies, the scientists at the Institute of Cell Therapy in Ukraine developed a novel method of treatment of pancreonecrosis using cord blood stem cells and umbilical cord tissue.
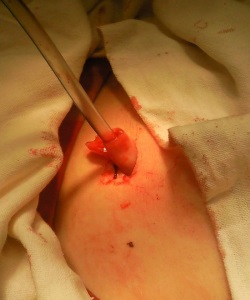 The treatment was tested in a randomized placebo-controlled clinical trial approved by the Ministry of Health of Ukraine (trial № Р-007/08 code Р-001.08) aimed to evaluate the efficacy of the umbilical cord blood stem cell drug Pancrostem. The clinical trial was preceded by preclinical studies in animals that showed the efficacy of this approach. The study lasted during 2008-2011 and enrolled 179 patients, aged from 20 to 80 (control group of 106 patients and Pancrostem-treated group of 73 patients) with aseptic pancreonecrosis, infected pancreonecrosis, and pancreatogenic abscesses.
The treatment was tested in a randomized placebo-controlled clinical trial approved by the Ministry of Health of Ukraine (trial № Р-007/08 code Р-001.08) aimed to evaluate the efficacy of the umbilical cord blood stem cell drug Pancrostem. The clinical trial was preceded by preclinical studies in animals that showed the efficacy of this approach. The study lasted during 2008-2011 and enrolled 179 patients, aged from 20 to 80 (control group of 106 patients and Pancrostem-treated group of 73 patients) with aseptic pancreonecrosis, infected pancreonecrosis, and pancreatogenic abscesses.
Pancrostem is simply the product name of allogeneic cord blood total nucleated cells (TNCs) that are applied as therapy for pancreatitis and pancreonecrosis. The Institute of Cell Therapy collects cord blood donations in utero with blood collection bags holding CPDA, erythrocytes were depleted by HES-sedimentation, and plasma was reduced by centrifugation. The isolated TNCs were cryopreserved with DMSO/Dextrane 40 solution in vials using a software-based freezer. Pancrostem was tested for quantitative and qualitative characteristics according to FACT-NetCord International Standards with some modifications. These units of donated and preserved cord blood TNCs constitute an off-the-shelf cell therapy product.
The Pancrostem clinical trial tested the standard management of pancreonecrosis that was described above against a combination of standard treatment plus an intravenous infusion of Pancrostem. Each pancreonecrosis patient received cord blood TNCs from a single donor, without HLA matching. The volume of umbilical cord blood TNCs (Pancrostem) delivered in each treatment was 20-50 ml, corresponding to a dose between 240 million and 1.2 billion TNCs per sample.The treatment with Pancrostem started on the third day following operative treatment (either surgery or drainage) and lasted five days. The control group received placebo instead of Pancrostem.
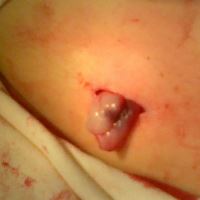 The umbilical cord tissue contributed to the pancreonecrosis treatment as part of the surgical care for patients. Contact between the abdominal wounds and the umbilical cord tissue stimulates reparative processes in the patient’s connective tissues. Cryopreserved whole cord tissue is also an off-the-shelf therapy product that was previously donated and cryopreserved. The cord tissue was not selected to come from the same donor as the cord blood. Sections of native cord tissue were also used as surgical grafts to close pancreatic fistulas and promote healing.
The umbilical cord tissue contributed to the pancreonecrosis treatment as part of the surgical care for patients. Contact between the abdominal wounds and the umbilical cord tissue stimulates reparative processes in the patient’s connective tissues. Cryopreserved whole cord tissue is also an off-the-shelf therapy product that was previously donated and cryopreserved. The cord tissue was not selected to come from the same donor as the cord blood. Sections of native cord tissue were also used as surgical grafts to close pancreatic fistulas and promote healing.
Results of the clinical trial showed that interventions with cord blood and cord tissue were correlated with statistically significant improvements in outcomes and survival for pancreatitis patients:
- The use of minimally invasive surgery under ultrasound guidance, followed by the infusion of Pancrostem, led to a 1.65 fold (р<0,05) increase in cured patients who were able to avoid open surgery. The duration of their hospital stay was shortened 1.47-fold (р<0,05) compared to the control group.
- The patients treated with Pancrostem who went on to a second stage of open surgery were able to postpone their surgery, which contributed to improved outcome.
- The overall mortality of Pancrostem-treated patients was 2.06-fold (р<0,05) lower compared to the control group, and their duration of post-operative hospital stay was 1.83-fold lower (р<0,05).
- During the five days of Pancrostem infusion, the early mortalityof study patients from enzymatic toxaemia was decreased 1.98-fold (р<0,05).
- Among patients with necrotic pancreatitis, those who received Pancrostem infusions and cord tissue grafts showed 4,71-fold (р<0,001) lower incidence of gastrointestinal bleeding from the abdominal cavity (omental bursa) compared to control group.
- The development of disseminated blood clotting syndrome in patients treated with Pancrostem occurred 3,48-fold (р<0,001) less often compared to the control group.
- In the retrospective literature, the incidence of pancreatic cysts in these patients usually reaches 30-50%, but in patients treated with Pancrostem the development of pancreatic cysts only occured in 6,1% of cases and the incidence of pancreatic fenestration was 3.12-fold lower (р<0,001) compared to the control group.
- The frequency of post-operative complications in the placebo-treated group made up 48.1% versus 24,7% in the Pancrostem-treated group.
To conclude, the obtained results demonstrate the effectiveness of the cryopreserved umbilical cord blood TNCs (Pancrostem) as part of the complex treatment of patients suffering from necrotic pancreatitis. The were no immune or allergic reactions registered. The use of the cell therapy significantly decreased the frequency of postoperative complications, improved overall survival, and hastened the recovery of patients with this difficult and often fatal condition.
As a result of this clinical trial, in 2012 the Ministry of Health of Ukraine approved Pancrostem as an accepted standard therapy for pancreonecrosis. Another cell therapy developed by the Institute of Cell Therapy was also approved in 2012 for critical lower limb ischemia. Meanwhile, ongoing clinical trials are testing four more therapies for cardiomyopathy, hepatitis, liver cirrhosis, and type 2 diabetes.



 Institute of Cell Therapy
Institute of Cell Therapy