You are here
From Bank to Bedside: CHA of South Korea
How big is the CHA Bio Group?
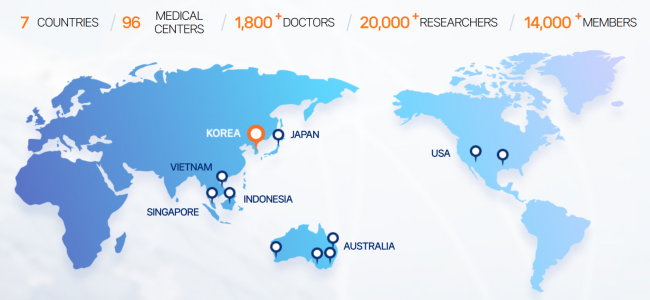
CHA Bio Group is a global life sciences group that includes publicly listed companies such as CHA Biotech and CMG Pharmaceutical, along with affiliates including CHA Healthcare, CHA Meditech, CHA Bio F&C, and CHA Care. The group has established Korea’s only integrated industry–academia–research–hospital ecosystem through CHA University, the CHA Advanced Research Institute (CHARI), and CHA Hospitals. It operates a disease-specific cell library that is one of the largest in the world, encompassing embryonic stem cells, adult stem cells (this includes cord blood), and immune cells. Based on this foundation, CHA conducts research and manufacturing of advanced biopharmaceuticals, including cell and gene therapies, while also expanding its third-generation cell and gene therapy CDMO business. In addition, CHA Bio Group operates one of the largest medical networks in the Asia-Pacific region, with 96 clinical centers across 7 countries and approximately 1,800 medical professionals.
What types of biobanking are available at CHA?
In 2025, CHA University, CHA Hospitals, and CHA Biotech were selected by the Government of the Republic of Korea as lead institutions for the “Global K-Cell Bank and Library (Specialized Cell Research Institute).” This initiative was established to strengthen global competitiveness in cell and gene therapy (CGT).
CHA possesses proprietary technologies and patents for generating somatic cell nuclear transfer (SCNT) stem cells, created by transferring a somatic cell nucleus into an enucleated oocyte. Currently, only a few institutions worldwide—including Columbia University, Oregon Health & Science University, and CHA—have this capability.
Global K-Cells are clinical-grade stem cells developed using Korea’s proprietary technologies. These cells can differentiate into all human cell types—including neurons, hepatocytes, cardiomyocytes, and blood cells—and serve as key raw materials for applications such as treatments for intractable diseases, artificial organs, and immune cell therapies.
CHA has also secured proprietary technologies for oocyte-derived parthenogenetic (haploid) stem cells, which are free from ethical concerns related to embryo destruction. In addition, CHA has established a comprehensive biobanking system covering the entire human life cycle. This includes cord blood, placenta, adipose-derived mesenchymal stromal cells (MSC), peripheral blood stem cells, Natural Killer (NK) cells, and skin cells.

CHA Biotech operates the family cord blood bank, iCord, which holds the largest asset base among cord blood banks in Korea, with approximately KRW 2 trillion in consolidated assets. The cord blood bank at CHA Biotech currently stores 82,245 private cord blood units and 8,069 public cord blood units. Collected cord blood undergoes rigorous testing and processing before being cryopreserved in liquid nitrogen at −196°C. With over 20 years of operational experience, CHA Biotech provides a fully integrated, end-to-end service, from cord blood storage to potential future transplantation.
What types of cell therapy research are developed at CHA hospitals?
 Dr. Minyoung Kim has led multiple clinical trials using cord blood-derived stem cells for cerebral palsy treatment. In 2010, the therapeutic effects of autologous cord blood stem cells were confirmed. In 2013, CHA demonstrated for the first time globally that allogeneic cord blood therapy significantly improved motor function in children with cerebral palsy. MRI findings showed increased white matter neural fiber density in treated patients. In 2015, further research into inflammation and immune responses revealed a reduction in inflammation in the periventricular region following cord blood therapy. Notably, patients who exhibited stronger immune responses showed greater improvements in motor function, leading to the first identification of the therapeutic mechanism of cord blood therapy for cerebral palsy.
Dr. Minyoung Kim has led multiple clinical trials using cord blood-derived stem cells for cerebral palsy treatment. In 2010, the therapeutic effects of autologous cord blood stem cells were confirmed. In 2013, CHA demonstrated for the first time globally that allogeneic cord blood therapy significantly improved motor function in children with cerebral palsy. MRI findings showed increased white matter neural fiber density in treated patients. In 2015, further research into inflammation and immune responses revealed a reduction in inflammation in the periventricular region following cord blood therapy. Notably, patients who exhibited stronger immune responses showed greater improvements in motor function, leading to the first identification of the therapeutic mechanism of cord blood therapy for cerebral palsy.
CHA Biotech is implementing differentiated development strategies for autologous and allogeneic cell therapies based on its integrated industry–academia–research–hospital ecosystem.
For autologous cell therapies, the company aims to expand patient access by leveraging Korea’s Advanced Regenerative Medicine and Advanced Biopharmaceuticals Act. The CHA pipeline of autologous cell therapy includes:
- Tumor-infiltrating lymphocyte (TIL) therapies for ovarian, head and neck, and gynecologic cancers.
- NK cell therapies for glioblastoma, triple-negative breast cancer, and hepatocellular carcinoma.
- Regulatory T-cell (T-reg) therapies for graft-versus-host disease (GVHD) and autoimmune diseases.
- Adipose-derived MSC therapies for premature ovarian insufficiency and endometrial diseases.
For allogeneic cell therapies, CHA Biotech is conducting research and development in collaboration with global partners, with the aim of technology out-licensing and entry into overseas markets. The CHA pipeline of allogeneic cell therapy includes:
- Umbilical cord-derived MSC therapies targeting ovarian aging, infertility, and frailty.
- Genetically engineered MSC therapies for pulmonary fibrosis and chronic obstructive pulmonary disease (COPD).
- CAR-NK cell therapies for solid tumors.
- Exosome therapies for corneal diseases, burns, and interstitial cystitis.
Furthermore, the company has validated its technological competitiveness by out-licensing its human embryonic stem cell-derived retinal pigment epithelium (RPE) therapy for dry age-related macular degeneration to Astellas Pharma.
How big is the medical tourism business at CHA?
 CHA Medical Centers welcomed 15,000 medical tourists last year, making it the largest in Korea in terms of scale. In particular, CHAUM, our longevity center, is widely known among international celebrities and high-profile individuals. CHA is particularly recognized for infertility and OB-GYN services.
CHA Medical Centers welcomed 15,000 medical tourists last year, making it the largest in Korea in terms of scale. In particular, CHAUM, our longevity center, is widely known among international celebrities and high-profile individuals. CHA is particularly recognized for infertility and OB-GYN services.
In addition, CHA operates Total Cell Clinic in Tokyo, Japan, a specialized regenerative medicine center that attracts international patients. The clinic provides cell therapies certified by Japan’s Ministry of Health, Labor and Welfare following rigorous evaluation of safety, efficacy, and medical and cell management systems. It has conducted over 10,000 cell therapy cases and operates its own cell processing center.
What is the impact of Korea’s new Regenerative Medicine Law?
Since the Advanced Regenerative Medicine and Advanced Bio Pharmaceuticals Safety and Support Act was revised in February 2025, patient access to cell and gene therapies in Korea has improved as the scope of these treatments has expanded. The revision also allows regenerative medical technologies that have shown safety and efficacy in clinical research to be approved as advanced regenerative treatments, so they can be used by patients that have severe or incurable conditions even before full regulatory approval. This kind of institutional improvement is expected to unlock previously unmet demand from patients who didn’t have access to these treatment opportunities before. However, the barriers remain high, and further enhancements will likely be necessary to keep pace with the global advancement of cell therapies.
CHA Biotech has received approval to conduct an advanced regenerative clinical trial using its NK cell therapy candidate, CHANK-101. CHA Bundang Medical Center will lead the study, targeting newly diagnosed glioblastoma patients who have completed surgery followed by chemoradiation. The plan is to isolate and expand each patient’s own NK cells, enhance their activity, and administer CHANK-101 in combination with the standard chemotherapy drug temozolomide to evaluate safety and efficacy.
In addition, CHA Biotech is planning to move forward with a multi-center advanced regenerative clinical research support program, which includes NK cell therapy for patients with recurrent glioblastoma and adipose-derived MSC therapy for patients with chronic pain. We’ve already secured sufficient data from Total Cell Clinic in Japan, and based on our years of experience in cell therapy, we expect that applying this expertise in Korea will allow us to establish these treatments relatively quickly.
What is the CHA vision for healthcare in the years to come?
 CHA Bio Group is developing an international bio-cluster, “Cell Gene Bioplatform” (CGB), in Pangyo 2nd Techno Valley. The facility will comprise 10 above-ground floors and 4 basement levels, with a total floor area of 66,115 square meters (approximately 20,000 pyeong). CGB is designed as an integrated platform for cell and gene therapies, covering the entire value chain, from R&D and clinical development to manufacturing and business development. Key components include a cell and gene therapy contract development and manufacturing organization (CGT CDMO), a comprehensive cell bank capable of storing a wide range of human cells — including stem cells, oocytes (eggs), and NK cells — and an open innovation center (K-BIO CIC) to support biotech startups and venture companies. The cluster is expected to serve as core infrastructure for advancing the broader cell and gene therapy ecosystem.
CHA Bio Group is developing an international bio-cluster, “Cell Gene Bioplatform” (CGB), in Pangyo 2nd Techno Valley. The facility will comprise 10 above-ground floors and 4 basement levels, with a total floor area of 66,115 square meters (approximately 20,000 pyeong). CGB is designed as an integrated platform for cell and gene therapies, covering the entire value chain, from R&D and clinical development to manufacturing and business development. Key components include a cell and gene therapy contract development and manufacturing organization (CGT CDMO), a comprehensive cell bank capable of storing a wide range of human cells — including stem cells, oocytes (eggs), and NK cells — and an open innovation center (K-BIO CIC) to support biotech startups and venture companies. The cluster is expected to serve as core infrastructure for advancing the broader cell and gene therapy ecosystem.
CHA aims to grow into a global leader in cell therapy. It has established a fully integrated ecosystem spanning research, clinical care, and manufacturing.
CHA also aims to reduce the cost of cell therapies to one-tenth of current levels.
Based on its proprietary technologies and cell banking capabilities, CHA is also developing next-generation longevity projects, with the goal of attracting patients from around the world to Korea for high-quality medical services.


