Usted está aquí
What your placenta can and can't do

Many expectant parents research their options for their child's cord blood - whether to donate it, privately bank it, or have it discarded as medical waste - but what about the placenta? Can it, too, be a valuable source of cells that one should consider saving? The answer is yes! First, it is important to know the role of the placenta in pregnancy.
The placenta is a maternal-fetal organ that develops from the fetus and attaches to the uterus during pregnancy. The placenta is responsible for, among other things, providing the growing fetus with oxygen and nutrients while at the same time removing toxins. The maternal side of the placenta carries blood from the uterus. The fetal side of the placenta receives the baby's blood from the umbilical cord. The maternal and fetal blood vessels are close enough within the placenta to allow for the transfer of nutrients, oxygen, and waste products; however, the two circulations do not mix. Once the baby is born, the placenta detaches from the uterine wall and is expelled from the body. The placenta is often referred to as "the afterbirth".
There's no doubt that the placenta is a unique and amazing organ, but to date, when it comes to stem cell banking, it often takes a back seat to cord blood. This is not surprising, as the first cord blood transplant occurred in Oct. 1988, whereas the first placental blood transplant took place nearly 20 years later in March of 2008.
Options for banking placental stem cells
Just as it is possible to bank umbilical cord blood and cord tissue, there are options available to bank blood and tissue from the placenta.
What is placental blood?
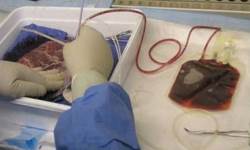 Placental blood is the blood that remains within the blood vessels of the placenta after the baby is born. It can be collected after the placenta is delivered. During pregnancy, the baby’s blood supply circulates through both the umbilical cord and the placenta, so that placental blood and cord blood are basically the same.
Placental blood is the blood that remains within the blood vessels of the placenta after the baby is born. It can be collected after the placenta is delivered. During pregnancy, the baby’s blood supply circulates through both the umbilical cord and the placenta, so that placental blood and cord blood are basically the same.
Placental blood contains hematopoietic stem cells (stem cells which make blood cells) like those found in the umbilical cord blood. The traditional application of cord blood is transplants where the patient’s blood and immune system need to be replaced with stem cells from a donor.Cord blood also has also been effective in clinical trials for neurologic conditions that occur in young children, such as cerebral palsy and autism.
Some researchers say that blood taken from the placenta contains a higher concentration of cells that are developmentally more immature, and holds more mesenchymal cells than blood in the umbilical cord. This is important because more stem cells are correlated with better treatment outcome, and more immature cells are less likely to invoke a graft-versus-host response in the transplant recipient.
Why preserve placental tissue?
 Placental tissue banking gives the opportunity to store stem cells that may have applications quite different from those of blood stem cells. Tissues from the placenta are an abundant source of mesenchymal stromal cells (MSCs), and these cells are being studied around the world for applications in regenerative medicine, due to their well-documented ability to reduce inflammation and modulate the immune system. The placenta also contains endothelial stem cells that can promote blood vessel formation, and epithelial stem cells that are precursors to the growth of skin.
Placental tissue banking gives the opportunity to store stem cells that may have applications quite different from those of blood stem cells. Tissues from the placenta are an abundant source of mesenchymal stromal cells (MSCs), and these cells are being studied around the world for applications in regenerative medicine, due to their well-documented ability to reduce inflammation and modulate the immune system. The placenta also contains endothelial stem cells that can promote blood vessel formation, and epithelial stem cells that are precursors to the growth of skin.
The history of the placenta in medicine goes back long before cord blood. Membranes of the placenta were documented as effective wound dressings in the early 1900’s. Placental tissue has been used for eye surgery since 1940. Within the past decade, clinical trials have studied the role of placental stem cells in wound healing, auto-immune disorders, arthritis, and sports medicine.
How should parents approach placenta banking?
Currently, at least two dozen organizations across the United States are asking mothers for placenta donations. Donating your placenta is free. Donated placentas supply the already approved medical therapies that utilize placental tissues and contribute to the development of new therapies.
More and more cord blood banks are offering parents private "placenta banking" for an additional fee. But almost all of the cord blood banks that make this claim are not actually extracting any stem cells from the placenta, they are just freezing a snippet of placenta tissue.
Also, verbal reassurances from sales representatives do not count, parents should seek reassurances in writing that are legally binding. Parents deserve to receive a final laboratory testing report on the banked product, just like the reports parents get when they bank cord blood stem cells.
Other choices
Lastly, there are traditions and fads that exist around the uses of the placenta. In most hospitals, parents can choose to take their placenta home with them after birth. However, you should check in advance to confirm this.
One practice, known as "Lotus Birth", is to leave the umbilical cord and placenta attached until they fall off. Bear in mind that the placenta is an organ containing about one pound of flesh and it will "go bad" long before it falls off.
Some mothers are eating their placenta, a choice which mimics the behavior of mammals in the wild. While the placenta is very nutritious, the process of cooking it or powdering it to be encapsulated in pills will destroy the hormones and nutrients that are unique to the placenta, as compared to other meats.
There are various ways to honor the placenta and its role in your baby’s birth. One practice is to bury the placenta and plant a tree over it. There are also companies that offer to make jewelry from your placenta. These options create long-lasting mementos of your child’s birth, but they also pass up the chance to store your child’s stem cells in the placenta.
Of course, no one should feel obligated to store their child's cord blood, cord tissue, placental blood, or placental tissue. If you decide this is not right for your family, please consider donating these valuable resources, so that others might benefit from the gift of this amazing organ.