Você está aqui
World Cord Blood Inventory 2012
According to the World Marrow Donor Association (WMDA) 2012 annual report, as of 1 Jan. 2012 the world inventory of cord blood in public banks was 591 thousand. By comparison, the Parent's Guide to Cord Blood Foundation finds that as of 31 Dec. 2012, the world inventory of cord blood in family banks was over 2.47 million.
The Parent's Guide to Cord Blood Foundation inventory is based upon confidential interviews with the management of family cord blood banks in our world database. The Parent's Guide to Cord Blood Foundation also maintains an accompanying database of case histories of patients who received therapy with cord blood stored in family banks, and the total number of cases is now close to one thousand. Information from the bank and patient databases are only released in aggregate formats, to preserve both the confidentiality of the bank business information (unless it is already public knowledge) as well as the privacy of the patient medical histories. The only person who has ever seen the raw contents of either database is Dr. Frances Verter, and the databases are stored on encrypted hard-drives. A report on the bank and patient data was presented as poster #63 at the 2013 annual ISCT meeting.
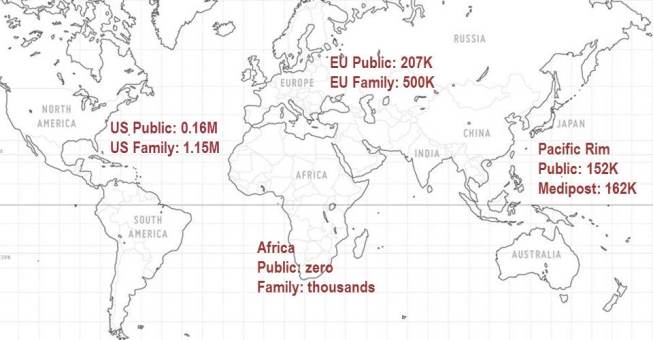
The map above shows some breakdowns of cord blood storage by geographic region. Our database is accurate for the regions of USA and EU, but incomplete for the regions of South America and Asia. Cord blood banking started in the USA, where the world's largest public bank is the NY Blood Center (current inventory over 60,000) and the world's largest family bank is Cord Blood Registry (current inventory 425,000). In the WHO European region, the WMDA reports a net inventory of 207,385 among public cord blood banks, whereas the family bank trade association Cord Blood Europe reports a net inventory of 500,000 among its member family cord blood banks as of Oct. 2012. Whereas the WMDA finds the net inventory in public cord blood banks in the WHO Western Pacific region to be 151,700, by comparison the largest mixed public/family bank in Korea, Medipost, exceeds that with an inventory of 162,429 as of Feb. 2013. According to our database, Medipost is currently the world's 5th largest cord blood bank of any type, and the largest outside of the US and EU. The only country in the world that has slightly more cord blood in public banks than in family banks is Japan, where both inventories are close to 30 thousand.
Obviously, the slow growth of public cord blood banking for unrelated transplants, which is usually dependent on government and charitable funding, is completely dwarfed by the successful business model of family cord blood banking as a form of health insurance. It is in the best interests of public health, at a global scale, to bring the operating standards of all family banks up to the public bank level and harness more of their storage for therapeutic purposes.
It has often been suggested that public cord blood banks should adopt a "hybrid" business model, in which the same bank offers both public and family banking, as a path to economic viability. Yet most cord blood banks around the world continue to operate predominantly in either the public or family mode of banking. Instead of economic forces driving bank practices, a new trend is that new clinical applications of cord blood are pushing banks towards hybrid behavior.
Consider clinical trials for cerebral palsy and similar acquired neurological conditions as an illustration of shifts in cord blood banking practices: The use of cord blood as therapy for these conditions was pioneered by Dr. Kurtzberg at Duke University Medical Center in 2004, and was initially only available to children who had their own cord blood privately banked. Since then, about 300 clients from family banks have gone to Duke to receive their own cord blood for acquired neurological conditions (Ref: PGCB patient database). But recently over 60 children have entered clinical trials (NCT00593242, NCT01445041) at Duke by relying on the university's public cord blood bank to provide personal storage for regenerative therapy. Meanwhile, in Korea, a couple of recent clinical trials (NCT01193660, NCT01528436) have treated over 50 children for cerebral palsy with matched donated cord blood units. In each of these cases a public bank is providing cord blood not for transplants but for regenerative therapy, in one case autologous and the other allogeneic.
Another example of changing paradigms can be seen in India, a country that does not have a national network of public cord blood storage, but does have a robust private banking sector. In recent years dozens of children have received sibling cord blood transplants for thalassemia, a treatment option that would not have been available to them without family cord blood banks.
The common theme in all of these cases is that cord blood bank practices are moving away from the silo concept that public banks only provide transplants and family banks mainly supply regenerative therapies. We are seeing more public banks facilitating regenerative therapy, and in some parts of the world patients must rely on family banks for transplants.


