Usted está aquí
Wound Healing with Hydrogels that hold Amniotic Fluid and Membrane
Extensive burns and full thickness skin wounds can be devastating to patients, even when treated. Delayed or inappropriate treatments can cause wide-ranging tissue scarring, often leading to impaired function. Additionally, the treatment of chronic wounds is very costly.
The gold standard of wound treatment currently used in clinics is a skin graft where patients undergo the removal of skin from a secondary site, which is then applied to the wound or burn. This therapy is limited, however, by the number of donor tissue sites, making it not feasible in the case of extensive or chronic, non-healing wounds. Donor tissue grafts are an additional option, but suffer from the need for immunosuppressive drugs to prevent graft rejection.
Limitations of autologous donor grafts have led to the development of bioengineered and synthetic wound dressings, which are most often comprised of a polymeric scaffold. Although such materials can improve wound healing, they are costly to produce and often result in relatively poor cosmetic outcomes.
Perinatal tissues, such as the amniotic membrane and amniotic fluid, have been shown to offer safe and effective grafts for wound care. Amniotic stem cells are known to secrete a wide spectrum of cell signaling molecules and growth factors that could have beneficial effects for wound healing. However the clinical application of amniotic membrane has been limited by the difficulty in handling and placing thin sheets of membrane, and the high costs associated with the use of whole living tissues.
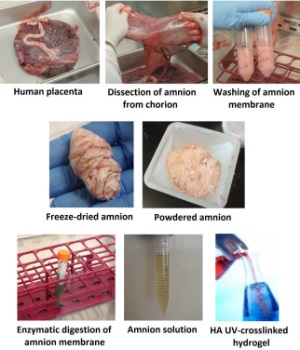 Our group at the Wake Forest Institute for Regenerative Medicine (WFIRM) seeks to overcome these obstacles and provide better wound healing. We report on two new advances that process perinatal cells and tissues so that they can be delivered within a liquid hydrogel. This method of delivery offers significant benefits in the ease of application and therapeutic effectiveness of the active cells.
Our group at the Wake Forest Institute for Regenerative Medicine (WFIRM) seeks to overcome these obstacles and provide better wound healing. We report on two new advances that process perinatal cells and tissues so that they can be delivered within a liquid hydrogel. This method of delivery offers significant benefits in the ease of application and therapeutic effectiveness of the active cells.
One advance developed at WFIRM is an amniotic membrane-derived hydrogel that is easy to apply and has proven effective in accelerating the healing of full-thickness wounds. Our novel processing method for the amniotic membrane, illustrated in the accompanying graphic, results in a liquid formulation that maintains its therapeutic properties, but without containing living cells. The combination of amniotic membrane cells with a liquid hydrogel provides an easy-to-use and off-the-shelf delivery system that releases therapeutic effects, while providing a protective and hydrating cover for wounds.
We have recently shown1 in pre-clinical animal studies that the amniotic membrane hydrogel accelerates the formation of new tissue for long-term wound closure, and regenerates thicker skin along with increased numbers of new blood vessels, compared to untreated or hydrogel carrier controls.
Another advance developed at WFIRM is the use of a 3D bioprinter to deposit living amniotic fluid stem cells directly into full-thickness wounds to accelerate healing. The printed stem cells are carried in a hydrogel that not only maintains cell survival and function during the printing process, but also supported the release and delivery of cell signaling molecules and growth factors by the stem cells. A pre-clinical study of this method was recently published2 and the 3D printing of stem cells is illustrated in the embedded YouTube video. The study showed that printed amniotic fluid stem cells accelerated wound healing accompanied by increased production of structural proteins and increased blood vessel formation and maturation, compared to untreated or hydrogel carrier controls.
Multiple preclinical studies have now been performed to investigate new and more effective ways to use perinatal tissues such as the amniotic membrane and fluid to improve wound healing. These studies are now being repeated in advanced preclinical studies and discussions with the FDA are underway to translate this research into clinical trials for patients with large or chronic wounds.
References
- Murphy SV, Skardal A, Song L, Sutton K, Huag R, Mack DL, Jackson J, Soker S, Atala A. Solubilized Amnion Membrane Hyaluronic Acid Hydrogel Accelerates Full-thickness Wound Healing. Stem Cells Translational Medicine 2017; 6(11):2020-2032.
- Skardal A, Murphy SV, Crowell K, Mack D, Atala A, Soker S. A tunable hydrogel system for long-term release of cell-secreted cytokines and bioprinted in situ wound cell delivery, J. Biomedical Materials Research 2016; 105(7):1986-2000.



 Oula Khoury is a PhD candidate at Wake Forest School of Medicine. She holds a Masters in Molecular Biology from the Lebanese American University in Beirut. Her PhD advisor Sean Murphy joined Wake Forest Institute for Regenerative Medicine in 2012 as a Postdoctoral Fellow and became an Assistant Professor in 2015. He obtained a Bachelors degree in Molecular Biology (Honors) from the University of Western Australia in 2006 and his PhD in Stem Cell Therapy in 2012. His research interests focus on applications of perinatal stem cells and tissues for lung disease and wound healing. He participates in a research team that is applying a multidisciplinary approach combining tissue engineering, extracellular matrix hydrogels, and 3D bioprinting to develop new treatment modalities.
Oula Khoury is a PhD candidate at Wake Forest School of Medicine. She holds a Masters in Molecular Biology from the Lebanese American University in Beirut. Her PhD advisor Sean Murphy joined Wake Forest Institute for Regenerative Medicine in 2012 as a Postdoctoral Fellow and became an Assistant Professor in 2015. He obtained a Bachelors degree in Molecular Biology (Honors) from the University of Western Australia in 2006 and his PhD in Stem Cell Therapy in 2012. His research interests focus on applications of perinatal stem cells and tissues for lung disease and wound healing. He participates in a research team that is applying a multidisciplinary approach combining tissue engineering, extracellular matrix hydrogels, and 3D bioprinting to develop new treatment modalities.