Jste zde
Omidubicel from Gamida Cell leads the way to Cord Blood Transplant version 2.0
 In May 2020, Gamida Cell announced preliminary results of their phase 3 trial of stem cell transplants with the expanded cord blood product Omidubicel (formerly known as NiCord®)1. A single cord blood unit expanded into Omidubicel provided transplant patients with neutrophil engraftment in a median of 12 days1,2. This is about 10 days faster than the 20-22 day engraftment baseline for single or double cord blood transplants2-4. Perhaps more important, it is even faster than the 16-19 day neutrophil recovery seen with various types of bone marrow transplants5,6. Throughout this article, when we say neutrophil recovery, it is defined as the time to reach an absolute neutrophil count (ANC) of 500 per microliter of blood. The faster engraftment achieved with Omidubicel can help make expanded cord blood products the new and improved, version 2.0, of cord blood transplants.
In May 2020, Gamida Cell announced preliminary results of their phase 3 trial of stem cell transplants with the expanded cord blood product Omidubicel (formerly known as NiCord®)1. A single cord blood unit expanded into Omidubicel provided transplant patients with neutrophil engraftment in a median of 12 days1,2. This is about 10 days faster than the 20-22 day engraftment baseline for single or double cord blood transplants2-4. Perhaps more important, it is even faster than the 16-19 day neutrophil recovery seen with various types of bone marrow transplants5,6. Throughout this article, when we say neutrophil recovery, it is defined as the time to reach an absolute neutrophil count (ANC) of 500 per microliter of blood. The faster engraftment achieved with Omidubicel can help make expanded cord blood products the new and improved, version 2.0, of cord blood transplants.
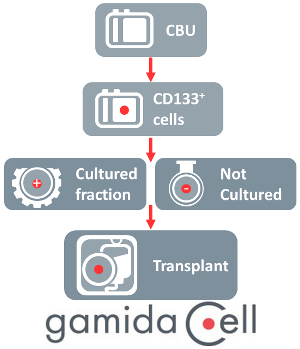
The journey to this achievement began well over a decade ago. Since 2007, Gamida Cell has run nearly a dozen clinical trials of both NiCord® for hematologic malignancies, and its sister product CordInTM for hemoglobinopathies7. Gamida Cell creates their expanded product by isolating CD133+ cells from a cord blood unit and then culturing them for 3 weeks with the small molecule nicotinamide and other growth factors. The molecule nicotinamide, a form of vitamin B-3, has been shown to inhibit the differentiation of stem cells, so that they keep multiplying as stem and progenitor cells. It also increases the ability of CD34+ progenitor cells to home to the bone marrow and engraft more efficiently8.
Gamida Cell’s recently completed phase 3 trial NCT02730299 was the largest trial to date of an expanded cord blood product. The clinical trial recruited 125 patients with various high-risk hematologic malignancies from over 50 hospitals in countries around the world.
Gamida Cell was also the first company to give an expanded cord blood product as a stand-alone graft, not as part of a double cord blood transplant with one expanded unit and one unmanipulated unit. Their first trial NCT01816230 of NiCord®/Omidubicel as a stand-alone graft treated 36 patients between 2013 and 2017 and has been published2. To be specific, when NiCord® is used as a “stand-alone” graft, it does not mean that the entire cord blood unit is expanded. Actually, after the CD133+ fraction of the mononuclear cells is selected, the remainder of the cord blood unit is cryopreserved. Then the CD133+ cells are cultured to create the NiCord®/Omidubicel product, which is also cryopreserved. Finally the two fractions of the original cord blood unit are delivered together to the treatment hospital to be thawed and transplanted.
What does the success of expanded cord blood mean for the practice of oncology? The cord blood community hopes it will lead to “a resurgence of cord blood” as a transplant graft choice9. The product NiCord®/Omidubicel has received several expedited designations from regulators: FDA Breakthrough Therapy Designation in 2016, European Medicines Agency Orphan Drug Designation in 2017, and FDA Orphan Drug in 201810-12. All of these designations acknowledge the important role that cord blood transplants play in providing donors for patients that cannot find suitable bone marrow donors, especially patients that belong to racial groups which are not well represented in the registries of adult donors13.
In the field of stem cell transplantation, there is a bit of dichotomy between the measures that are important to oncologists versus the measures that matter to health insurance companies. Oncologists aim to optimize the long term survival of their patients. The measures they track are Overall Survival (OS), Progression Free Survival (PFS), and non-relapse mortality (NRM). By these measures of survival, cord blood has repeatedly come out as either the best graft choice or comparable to other choices14-16.
By comparison, health insurance companies are primarily focused on the cost of different types of stem cell transplants. A RAND Corporation report estimated that in 2014 the average hospital charges for a stem cell transplant from an unrelated donor (and duration of hospital stay) was $485,000 (38 days) for bone marrow, $571,000 (34 days) for peripheral blood stem cells, and $831,000 (51 days) for cord blood17. The biggest factor contributing to the higher cost of cord blood transplants was the longer time to engraftment, which translates into more days in intensive care and more complications from infections. Gamida Cell has removed that disadvantage by driving down the engraftment time of a cord blood transplant so that it is competitive with bone marrow and peripheral blood transplants.
Based on their success to date, Gamida Cell has registered an expanded access clinical trial NCT04260698 of Omidubicel for patients with hematologic malignancies, with an estimated start date of 15 June 2020. At present in the United States, about 40% of patients that are eligible for a stem cell transplant do not receive one for various reasons, including the lack of a matched donor1. These patients might become a segment of the transplantation market that is served by expanded cord blood products like Omidubicel. At present nearly 80% of oncologists have little experience transplanting cord blood grafts; hopefully the faster engraftment of expanded cord blood products will help change that demographic18.
Four other companies also have expedited regulatory designations for expanded cord blood products19. Results have recently been presented for products from Magenta Therapeutics20, which expands cord blood with the aryl hydrocarbon receptor StemRegenin-1, and from ExCellThera21, which expands cord blood with the small molecule UM171. Both of these companies may have advantages over Gamida Cell in the ease and speed of manufacturing their expanded products. It will take years to evaluate the relative pros and cons of different expanded cord blood products, looking at factors such as manufacturing feasibility, frequency of failures to engraft, and comparisons of long-term outcomes.
For now, the bottom line is that expanded cord blood works. We are entering a second generation of cord blood transplants, using products that will hopefully become a routine option for stem cell transplant patients.
References:
- Gamida Cell. Gamida Cell Announces Positive Topline Data from Phase 3 Clinical Study of Omidubicel in Patients with High-Risk Hematologic Malignancies. Press Release 2020-05-12
- Horwitz ME, Wease S, Blackwell B, Valcarcel D, et al. Phase I/II study of stem-cell transplantation using a single cord blood unit expanded ex vivo with nicotinamide. J Clinical Oncology 2019; 37(5):367-374.
- Barker JN, Weisdorf DJ, DeFor TE, ... Wagner Jr. JE. Transplantation of 2 partially HLA-matched umbilical cord blood units to enhance engraftment in adults with hematologic malignancy. Blood 2005; 105(3):1343–1347.
- Milano F, Rezvani AR, Kurtzberg J, ... Delaney C. No Engraftment Advantage after Single or Double Umbilical Cord Blood Transplant (CBT) with the Addition of a Non-HLA Matched Off-the-Shelf Expanded Cord Blood Unit Compared to Conventional CBT: Results of a Randomized Trial. Blood 2019; 134(S1):146.
- Eapen M, Rubinstein P, Zhang M-J, et al. Outcomes of transplantation of unrelated donor umbilical cord blood and bone marrow in children with acute leukaemia: a comparison study. Lancet 2007; 369(9577):1947-1954.
- Bashey A, Zhang M-J, McCurdy SR, ... Eapen M. Mobilized Peripheral Blood Stem Cells Versus Unstimulated Bone Marrow As a Graft Source for T-Cell–Replete Haploidentical Donor Transplantation Using Post-Transplant Cyclophosphamide. J Clinical Oncology 2017; 35(26):3002–3009.
- Verter F, Bersenev A, and Couto PS. Clinical Trials of Expanded Cord Blood. Parent’s Guide to Cord Blood Foundation Newsletter May 2018
- Peled T, Shoham H, Aschengrau D, et al. Nicotinamide, a SIRT1 inhibitor, inhibits differentiation and facilitates expansion of hematopoietic progenitor cells with enhanced bone marrow homing and engraftment.Experimental Hematology 2012; 40(4):342-355.
- Eapen M. A Resurgence of Cord Blood Transplantation? Lancet Haematology 2020; 7(2):e89-e90.
- Gamida Cell. Gamida Cell Receives FDA Breakthrough Therapy Designation For NiCord®. Press Release 2016-10-11
- Gamida Cell. Gamida Cell Receives Additional EMA Orphan Drug Designation for NiCord® in Haematopoietic Stem Cell Transplantation (HSCT). Press Release 2017-03-23
- Gamida Cell. Gamida Cell Receives Orphan Drug Designation from the FDA for NiCord® as a Treatment for Hematopoietic Stem Cell Transplantation. Press Release 2018-07-17
- Barker JN, Boughan K, Dahi PB, ... Politikos I. Racial disparities in access to HLA-matched unrelated donor transplants: a prospective 1312-patient analysis. Blood Advances 2019; 3(7):939–944.
- Milano F, Gooley T, Wood B, ... Delaney C. Cord-Blood Transplantation in Patients with Minimal Residual Disease. NEJM 2016; 375:944-953.
- Ando T, Tachibana T, Tanaka M, et al. Impact of graft sources on immune reconstitution and survival outcomes following allogeneic stem cell transplantation.Blood Advances 2020; 4(2):408–419.
- Brunstein CG, Eapen M, Antin JH, et al. Results of Blood and Marrow Transplant Clinical Trials Network Protocol 1101 a Multicenter Phase III Randomized Trial of Transplantation of Double Umbilical Cord Blood Vs. HLA‐Haploidentical ‐Related Bone Marrow for Hematologic Malignancy. TCT Abstract 2020;
- Kapinos KA, Briscombe B, Gracner T, et al. Public Cord Blood Banks: Worthy of National Investment. RAND Corporation 2017.
- Barker J. On behalf of the ASTCT Cord Blood Special Interest Group. Cord Blood Transplantation in the Era of COVID: Challenges and Opportunities. HRSA Advisory Council on Blood Stem Cell Transplantation. Presentation 20202-04-27
- CellTrials.org FDA Expedited Approvals of Expanded Cord Blood Products. CellTrials.og Blog 2019-09-09
- Stefanski H, Brunstein CG, McKenna DH, ... Wagner Jr. JE. MGTA-456, an Aryl Hydrocarbon Receptor (AHR) Antagonist Based Expansion of CD34+ Hematopoietic Stem Cells (HSC), Permits Selection of Better HLA Matched Cord Blood Units (CBUs) and Promotes Faster Neutrophil Recovery and Uniform Engraftment with Potentially Less Acute Graft-Vs-Host Disease (GVHD). Blood 2019; 134(S1):804.
- Cohen S, Roy J, Lachance S, et al. Hematopoietic stem cell transplantation using single UM171-expanded cord blood: a single-arm, phase 1–2 safety and feasibility study. Lancet Haematology 2020; 7(2):E134-E145.