Jste zde
Cell Therapy Clinical Trials for AutoImmune Diagnoses 2011-2015
We are collaborating on a project to compile all cell therapy clinical trials registered worldwide from 2011 through 2015. Our database includes 58 clinical trials for autoimmune diseases, with a total planned enrollment of 2192 patients.
In this article we have prepared two figures to illustrate the importance of cell therapy clinical trials for autoimmune diseases and the variety of cell types currently in clinical trials for these diagnoses.
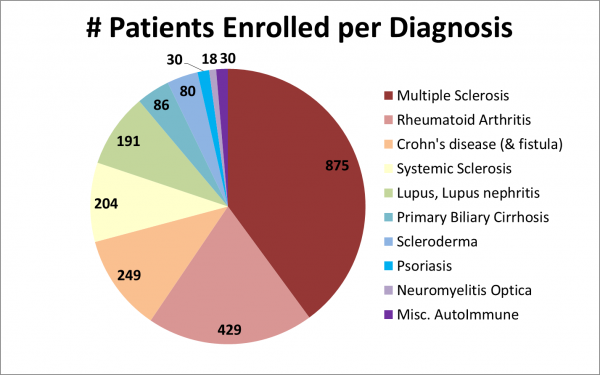
Figure 1: In this pie chart, the width of the pie slices is the number of patients that the clinical trials aim to enroll, and slices are color-coded by autoimmune diagnosis. The most common diagnosis is Multiple Sclerosis at 40% of patient enrollment.
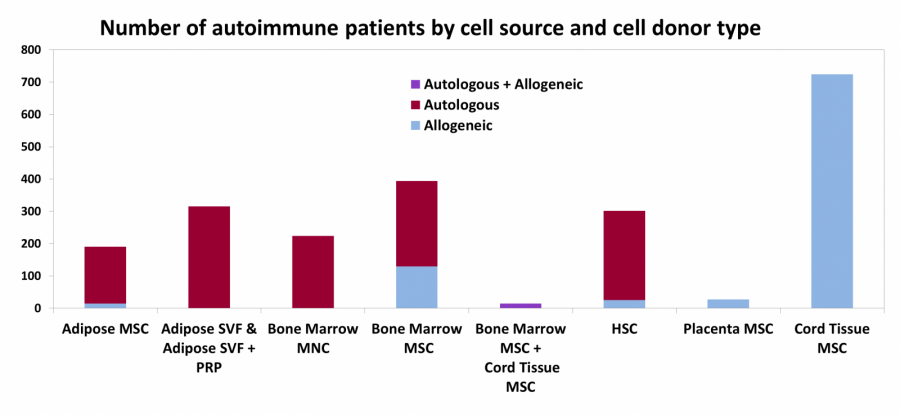
Figure 2: In this bar graph, the height of each bar is the number of patients that the clinical trials aim to enroll. There are separate bars for various types of cell sources, and within each bar they are color-coded by whether the source is autologous or allogeneic. In this graph 1256 (57%) patients are receiving autologous cell therapy and 1351 (62%) patients are receiving MSC therapy.